UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 8-K
CURRENT REPORT
Pursuant to Section 13 or 15(D)
of the Securities Exchange Act of 1934
January 8, 2020
Date of report (Date of earliest event reported)
Agile Therapeutics, Inc.
(Exact name of registrant as specified in its charter)
| Delaware | 001-36464 | 23-2936302 | |
| (State or other jurisdiction of incorporation) |
(Commission File Number) |
(IRS Employer Identification No.) | |
| |||
|
101 Poor Farm Road Princeton, New Jersey (Address of principal executive offices) |
08540 (Zip Code) | ||
|
Registrant's telephone number, including area code (609) 683-1880
(Former name or former address, if changed since last report)
|
Check the appropriate box below if the Form 8-K is intended to simultaneously satisfy the filing obligation of the registrant under any of the following provisions:
¨ Written communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425).
¨ Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12).
¨ Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b)).
¨ Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c)).
Securities registered pursuant to Section 12(b) of the Act:
| Title of each class | Trading symbol | Name of exchange on which registered |
| Common stock, par value $0.0001 per share | AGRX | The Nasdaq Capital Market |
Indicate by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (§230.405 of this chapter) or Rule 12b-2 of the Securities Exchange Act of 1934 (§240.12b-2 of this chapter).
Emerging growth company ¨
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ¨
Item 2.02 Results of Operations and Financial Condition.
As discussed below, in connection with its participation in the 38th Annual J.P. Morgan Healthcare Conference in San Francisco, California beginning on January 13th, 2020, Agile Therapeutics, Inc. (the “Company”) updated its corporate presentation to include disclosure that the Company had an estimated $34.5 million of cash and cash equivalents as of December 31, 2019, which includes the proceeds of approximately $19.3 million, net of expenses, from the sale of approximately 10.4 million shares of the Company’s common stock, par value $0.0001 per share (“Placement Shares”), representing the full amount of shares authorized for sale by the Company in the at-the-market offering conducted pursuant to the Common Stock Sales Agreement dated November 8, 2019 (the “Sales Agreement”) with H.C. Wainwright & Co., LLC. The issuance and sale of the Placement Shares by the Company under the Sales Agreement was made pursuant to a prospectus supplement to the Company’s registration statement on Form S-3, originally filed with the Securities and Exchange Commission (the “SEC”) on November 2, 2018, and declared effective by the SEC on November 14, 2018. The Company believes its estimated cash and cash equivalents as of December 31, 2019 will be sufficient to meet its projected operating requirements into September 2020.
Because the Company’s financial statements for the year ended December 31, 2019 have not yet been finalized or audited, the preliminary statement of the Company’s cash and cash equivalents as of December 31, 2019 in this Item 2.02 is subject to change, and the Company’s actual cash and cash equivalents as of the end of this period may differ materially from this preliminary estimate. Accordingly, you should not place undue reliance on this preliminary estimate.
Item 8.01. Other Events.
Beginning on January 13, 2019, the Company will participate in the 38th Annual J.P. Morgan Healthcare Conference in San Francisco, California. The Company has updated its corporate presentation that it intends to use in connection with its presentation at the Biotech Showcase 2020 on Tuesday January 14, 2020 at 10:00 a.m. Pacific Time, at the J.P. Morgan 2020 Healthcare Conference on Thursday, January 16, 2020 at 10:30 a.m. Pacific Time, and in meetings with investors. The updates primarily involve disclosure regarding the Company’s cash and cash equivalents as of December 31, 2019 as well as the Company’s plans for commercialization upon the approval of AG200 (Twirla®).
A copy of the Company’s corporate presentation is attached hereto as Exhibit 99.1 and is hereby incorporated by reference herein.
| Item 9.01. | Financial Statements and Exhibits. |
(d) Exhibits.
|
Exhibit |
Description | |
| 99.1 | Agile Therapeutics, Inc. Presentation |
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, as amended, the registrant has duly caused this report to be signed on its behalf by the undersigned hereunto duly authorized.
| Agile Therapeutics, Inc. | ||
| Dated: January 8, 2020 | By: | /s/ Alfred Altomari |
| Name: Alfred Altomari | ||
| Title: Chairman and Chief Executive Officer | ||
Exhibit 99.1

NASDAQ: AGRX Low - dose, Weekly Contraceptive Patch $3.8B Addressable Market

NASDAQ: AGRX Forward Looking Statement Certain information contained in this presentation and other matters discussed today or answers that may be given in response to questions may include “forward - looking statements” . We may, in some cases, use terms such as “predicts,” “believes,” “potential,” “continue,” “estimates,” “anticipates,” “expects,” “plans,” “intends,” “may,” “could,” “might,” “will,” “should” or other words that convey uncertainty of future events or outcomes to identify these forward - looking statements . In particular, the Company’s statements regarding projections and potential future results are examples of such forward - looking statements . The forward - looking statements are subject to important factors, risks and uncertainties, including, but not limited to, risks related to our ability to adequately respond to the deficiencies in the second Twirla CRL issued by the FDA on December 21 , 2017 , the potential that the FDA determines that our data do not support approval of the Twirla NDA and requires us to conduct additional studies or reformulate Twirla to address the concerns raised in the 2017 CRL, our ability to obtain and maintain regulatory approval of Twirla, the inability of our third - party manufacturer, Corium International, Inc . (Corium), to complete any work or provide any data and other information necessary to support the approval of our Twirla NDA, our ability along with Corium to complete successfully the scale - up of the commercial manufacturing process for Twirla, including the qualification and validation of equipment related to the expansion of Corium’s manufacturing facility, the performance and financial condition of Corium or any of the suppliers to our third - party manufacturer, the success and timing of our clinical trials or other studies, regulatory and legislative developments in the United States and foreign countries, our ability to obtain and maintain intellectual property protection for Twirla, our inability to timely obtain from our third - party manufacturer, Corium, sufficient quantities or quality of our product candidates or other materials required for a clinical trial or other tests and studies, our ability to raise capital when needed to complete the commercial launch of Twirla, and unforeseen market factors or events in our clinical, regulatory and manufacturing development plans ; and other factors, including general economic conditions and regulatory developments, not within the Company’s control . These factors could cause actual results and developments to be materially different from those expressed in or implied by such statements . The forward - looking statements are made only as of the date of this presentation and the Company undertakes no obligation to publicly update such forward - looking statements to reflect subsequent events or circumstance . For additional information about the risks and uncertainties that may affect our business please see the factors discussed in “Risk Factors” in the Company’s Annual Report on for 10 K for the year ended December 31 , 2018 and in the quarterly reports filed with the SEC . 2

NASDAQ: AGRX ▪ A champion for healthcare choices women deserve, headquartered in Princeton, NJ ▪ Dedicated to building a robust Women’s Health Franchise ▪ Twirla ® is our initial opportunity Weekly Contraceptive Patch $3.7B Addressable Market 3

NASDAQ: AGRX ▪ Short - Term Goal - Establish Agile in the prescription contraceptive market with Twirla, our lead product candidate ▪ Long - Term Mission - Broaden our women’s health portfolio in areas of unmet medical need 4 Agile’s Corporate Strategy: Become a Leader in Women’s Health Establish Agile in Contraceptive Market with Lead Product Candidate Become Contraceptive Market Leader Broaden Women’s Health Portfolio in Areas of Unmet Need

NASDAQ: AGRX 5 Agile’s Women’s Health Mission Starts with Contraception ~50% of pregnancies in U.S. women are unintended 3 Nearly half of unintended pregnancies are due to inconsistent and/or improper use of contraception 4 Women’s individual preferences for contraceptive methods vary and change across their lifetimes as their needs change 5 WHY CONTRACEPTION? WHY DO WOMEN NEED MORE BIRTH CONTROL OPTIONS? Women use contraception for an average of 30 years, and nearly all women use contraception at some point 1,2 Women are more consistent with contraceptive use and stay with a method for longer when using a method of their choosing 4 1 - Hamilton BE, Kirmeyer SE., National Center for Health Statistics. 2017; 2 - Daniels K et al, National Center for Health Statistics. 2013 3 - Finer LB and Zolna MR, NEJM 2016; 4 - Frost JJ and Darroch J., Perspectives on Sexual and Reproductive Health 2008 5 - Mansour D, Int J Women’s Health 2014

NASDAQ: AGRX Potential to reduce burden associated with daily pills 49% of contraception users prefer non - daily method 3 52% are frustrated with taking the pill daily 3 NON - DAILY OPTIONS Preferred by some women 4 Some women prefer to avoid injections, implants, and intrauterine devices LESS INVASIVE METHODS LOWER ESTROGEN DOSE The dose of estrogen in CHCs is believed to be the primary factor influencing the risk of blood clots (VTEs) 1 The only non - daily transdermal patch currently available delivers a high dose of estrogen 2 “ Some women are just not good at remembering to take a pill at the same time every day…Others don’t want something in their vagina while others don’t want an injection .” - Ob/Gyn 1 - Stegeman B. H., et al, BMJ 2013; 2 - Xulane Package Insert; 3 - Mansour D., International Journal of Women’s Health 2014; 4 - Quali tative and quantitative HCP research, Kantar Health 2010; Third party research, 2017 6 What is Missing From Available Hormonal Birth Control Options?

NASDAQ: AGRX ~ 30 µg/day Ethinyl Estradiol (EE) 120µg/day Levonorgestrel (LNG) 7 Twirla Designed to Fill A Hormonal Birth Control Market Need Less invasive than some methods (vaginal ring, IUDs, injections, implants) “I want to eliminate the forgetfulness… but I don’t want to lose that control either.” – Consumer, October 2016 Source: Qualitative consumer market research, Adelphi Research 2016 Pill Regimen: Once - a - day Patch Regimen: Once - a - week NON - DAILY LESS INVASIVE HORMONE PROFILE

NASDAQ: AGRX 8 U.S. Hormonal Contraceptive Market is a Significant Opportunity US Market Estimates (2018) $1.6B $5.3 Billion U.S. Contraceptive Market Combined Hormonal Contraception (CHC) Progestin - Only (P - Only) Long Acting Reversible Contraception (LARC) CHC Pill, Ring, Patch P - only Pill, Injection IUD, Implant $3.7 Billion $300 Million $1.3 Billion CHC Pills + Ring + Patch = $3.7 Billion Potential Addressable Market for Rings (18%) Patches (5%) IUDs (16%) Implants (8%) Injectables (5%) P - Only Pills (1%) CHC Pills (48%) P - only Pills category includes emergency contraceptive prescriptions. Not Shown: “All Other” category with <1M TRx Sources: IQVIA NSP through Dec 2018; ACOG FAQs

NASDAQ: AGRX 9 Twirla has the Potential for Significant Market Share Peak TRx Share Estimate Based on Consumer & Physician Market Research and Market Analogs Sources: IMS NPA, 2002 - 2014 Qualitative and Quantitative HCP and Consumer market research, Adelphi Research 2016 Quantitative HCP market research, MarketVision Research 2019 6 - 9% HCP Market Research (% CHC Market TRx) Study Year Stated Share Calibrated for Overstatement 2019 20% 14% 2016 23% 14% Average of Analog Brands 9.6% Consumers “Extremely Likely” to Ask for Twirla 15%

NASDAQ: AGRX 10 Twirla Pivotal Phase 3 SECURE Trial STUDY DESIGN Contemporary Study Design with Inclusive Patient Population RESULTS ▪ Frequent pregnancy testing ▪ Stringent cycle exclusion ▪ No exclusions for weight or BMI ▪ SECURE trial population, one of the heaviest for a CHC trial ▪ Tight confidence interval achieved on overall results ▪ Pearl Index higher than previously approved products ▪ Effect of obesity observed ▪ Low levels of detachments, patch site irritation, and itching ▪ Adhesion performance improved over time with increased use ▪ Observed learning curve may be addressable through training ▪ Low rate of hormone - related adverse events ▪ Serious adverse events observed in 1.97% of subjects ▪ Bleeding profile similar to low - dose oral CHCs Rigorous Design Representative of Women Seeking Hormonal Contraception Efficacy Safety and Tolerability Wearability Single - arm 13 - cycle trial of ~2000 healthy subjects aged ≥ 18 at 102 U.S. clinical sites

NASDAQ: AGRX 11 Recent Regulatory History Dec. 2017 Complete Response Letter (CRL) from FDA June 2018 Formal Dispute Resolution (FDR) on Adhesion Initiated Oct. 2018 Resolution of FDR & Potential Path Forward for Resubmission 2018 2020 Feb. 2019 Completion of Comparative Wear Study – Primary Endpoint Achieved 2019 Oct 2019 FDA Advisory Committee Meeting Votes 14 - 1 - 1 in Favor of Approval May 2019 New Drug Application (NDA) Submitted & Accepted PDUFA GOAL DATE: February 16, 2020

NASDAQ: AGRX CLARIFICATION FROM FDA FOR NEXT STEPS New method appears reasonable and will be a review issue Responses to initial PAI submitted. Likely subject to another PAI FDR completed; FDA recommended Comparative Wear Study with Xulane ® FDA anticipates reviewing efficacy and benefit/risk at Advisory Committee ; 12 The Path To Potential Twirla Approval ; ; ; ; ; ; ; ISSUE RAISED BY FDA CRL Adhesion Test Methods Manufacturing Inspection Observations In Vivo Adhesion High Pearl Index Achieved Primary Endpoint in Comparative Wear Study & Demonstrate Non - Inferior Adhesion to Xulane ® Completed NDA Submission to FDA Completed PAI at Corium Favorable FDA Advisory Committee on Efficacy and Benefit/Risk REGULATORY MILESTONES ACHIEVED TO DATE PDUFA GOAL DATE: February 16, 2020

NASDAQ: AGRX MARKET ACCESS ▪ Agile knows its market ▪ Top 8 payers expected to cover majority of commercial lives 1 ▪ Strategic contracting to place Twirla in competitive reimbursement position SALES FORCE ▪ Small, targeted sales force (70 - 100 reps) to launch ▪ Phased hiring linked to formulary acceptance ▪ Focus on high - prescribing Ob/ Gyns and women’s health NP/PAs MANUFACTURING ▪ Corium is an experienced contract patch manufacturer ▪ Qualification of commercial scale equipment in final stages ▪ Plan for completion of validation after approval LAUNCH NETWORK ▪ Relationships with experienced vendors to facilitate commercial launch ▪ Vendors have expertise in marketing, PR, market access, and supply chain Sources 1 - Berchick E, Hood E, Barnett J,. Health Insurance Coverage in the United State 2017 Haefner, M. America's largest health insurers in 2018 13 Agile Has Activated Partners to Prepare for Twirla Commercial Readiness

NASDAQ: AGRX Initiate consumer campaign 14 Phased Approach to Commercial Strategy Pre - PDUFA PRE - MARKET PREP Set up trade distribution Plan payer strategy Marketing Analytics & Strategy Launch manufacturing Payer strategy Formulary access Hire & train sales force Launch sales force Implement HCP Campaign Samples Availability PRODUCT ACCESS HCP ENGAGEMENT CONSUMER ACTIVATION Pre - Validation Validation Commercial Launch Manufacturing Preparations

NASDAQ: AGRX 0 10 20 30 40 50 60 70 ESI CVS OptumRx UHC Anthem Aetna Kaiser Cigna HCSC MedImpact Prime Thx Humana BCBS-MI BCBS-CA BCBS-FL Highmark Carefirst Emblem BCBS-MA HealthNet Top 20 Commercial Payers (MM Lives) PBM National Insurer Regional Insurer Managed Care Strategy: Minimize Access Barriers 15 ~ 75% of Commercial Lives Sources 1 - Berchick E, Hood E, Barnett J,. Health Insurance Coverage in the United State 2017 Haefner, M. America's largest health insurers in 2018 Strategy Will Inform Sales Force Rollout Plan Twirla ® Managed Care Strategy Patch Replacement Program Pricing Strategy Affordable Care Act State Mandates

NASDAQ: AGRX 16 AGRX Financial Overview $34.5 Million cash on hand at December 31, 2019 (Unaudited) ▪ The Company believes its estimated cash and cash equivalents as of December 31, 2019 will be sufficient to meet its projected operating requirements into September 202 0 $100.0 Million Universal Shelf Registration Statement on Form S - 3 ▪ In 2019, $42.3 million in total net proceeds raised through a private placement, public offering and our “At - the - Market” program offerings. ▪ 69.8 Million common shares outstanding at December 31, 2019. Capital Balance Sheet

NASDAQ: AGRX WHAT IS OUR INITIAL FOCUS? ▪ A non - daily, less invasive contraceptive option – Twirla ® ▪ Expect to launch into $3.7B addressable market ▪ 6 - 9% peak TRx market share estimate WHY ARE WE WELL - POSITIONED TO EXECUTE OUR PLAN? ▪ Experienced management team with the ability to plan and execute ▪ Activated partners to prepare for commercial readiness ▪ Building out internal capabilities WHAT DID WE SUCCESSFULLY COMPLETE IN 2019? 17 Summary of Agile Therapeutics Opportunity 2019 MILESTONE COMPLETION DATE Conduct Comparative Wear Study & Meet Primary Endpoint Completed Feb 2019 Pre - Approval (PAI) Inspection at Manufacturer Completed Oct 2019 Advisory Committee on Twirla Completed Oct 2019 WHAT’S NEXT FOR 2020? 2020 MILESTONE PROJECTED DATE Twirla PDUFA Goal Date February 16, 2020 Initial Shipment of Product 2H 2020

NASDAQ: AGRX 18 Appendix

NASDAQ: AGRX Evidence of Efficacy in a Representative Population 19 BMI Category BMI (kg/m 2 ) % of Study Population Pearl Index UB 95% CI Normal * < 25 39% 3.03 4.62 Overweight ≥ 25 - < 30 25% 5.36 7.98 Obese ≥ 30 35% 6.42 8.88 Non - Obese * < 30 65% 3.94 5.35 Obese ≥ 30 35% 6.42 8.88 ITT = Intent to Treat; all results shown are based on ITT subjects ≤ 35 years of age; UB 95% CI = upper bound of the 95% con fid ence interval Source: 1 Personal correspondence with Dr. James Trussell Population (ITT) Pearl Index UB 95% CI ≤ 35 years of age 4.80 6.06 *Reflective of Historical CHC Trial Populations A tight confidence interval was achieved on the overall results: Prespecified analyses showed an effect of obesity: Pearl Index with no contraception 1 : ~ 190 # On - treatment pregnancies # Cycles x 1300 Pearl Index =
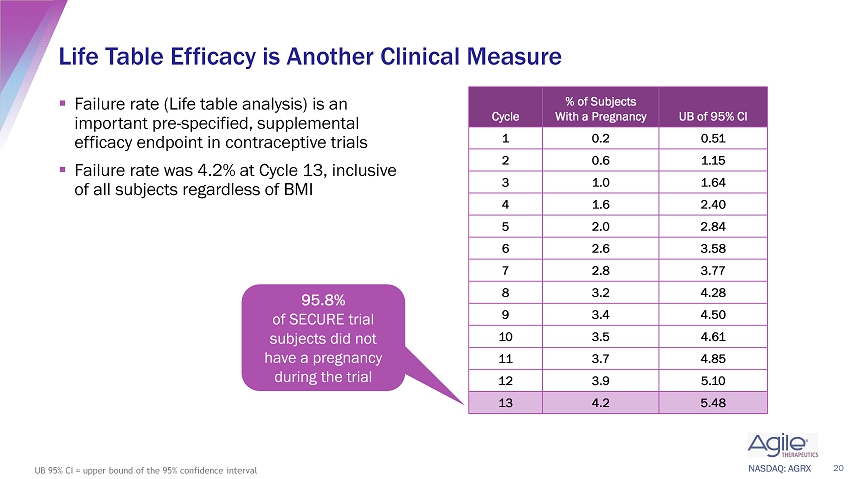
NASDAQ: AGRX ▪ Failure rate (Life table analysis) is an important pre - specified, supplemental efficacy endpoint in contraceptive trials ▪ Failure rate was 4.2% at Cycle 13, inclusive of all subjects regardless of BMI Life Table Efficacy is Another Clinical Measure 20 UB 95% CI = upper bound of the 95% confidence interval 95.8% of SECURE trial subjects did not have a pregnancy during the trial Cycle % of Subjects With a Pregnancy UB of 95% CI 1 0.2 0.51 2 0.6 1.15 3 1.0 1.64 4 1.6 2.40 5 2.0 2.84 6 2.6 3.58 7 2.8 3.77 8 3.2 4.28 9 3.4 4.50 10 3.5 4.61 11 3.7 4.85 12 3.9 5.10 13 4.2 5.48

NASDAQ: AGRX ▪ Overall serious adverse events (SAEs) were observed in 1.97% of the SECURE trial study population; generally in line with those observed in other low - dose combined hormonal products* (rate in Quartette trial = 1.6%); 0.7% of subjects had SAEs that were considered potentially study drug related, including deep vein thrombosis (DVT), pulmonary embolism (PE), gallbladder disease, ectopic pregnancy, and depression ▪ In the combined safety database for Agile Phase 3 trials (n >3,000), there were 5 subjects with potentially study drug related DVTs or PEs, 4 of whom were obese (BMI >30kg/m2) Safety and Tolerability Profile in the Phase 3 Trials 21 Low rates of hormone - related adverse events, consistent with publicly available information for other low - dose combined hormonal products: *Information is based on currently marketed product labels and publicly available information; adverse event (AE) terms utili zed in table (except nausea) represent composites of relevant specific AE preferred terms. Different terminology may be used in product labels and reports. We have not performed a head - to - head comparison of Twirla to Ortho Evra or Quartette. Adverse Event* SECURE Trial Prior Agile Phase 3 Trials Ortho Evra Trials* Quartette Trial* Total in Safety Population 2031 1043 3322 3597 Headache 4.5% 3.7% 21.0% 12.2% Nausea 4.1% 4.3% 16.6% 6.7% Breast tenderness/pain/discomfort 2.0% 1.8% 22.4% 2.2% Mood swings/changes/depression 2.7% 2.8% 6.3% 2.9% Heavy/irregular vaginal bleeding 2.6% 2.1% 6.4% 9.7%

NASDAQ: AGRX ▪ Bleeding - related side effects are among the top reasons women discontinue hormonal contraception 1 ▪ Monthly withdrawal bleeding duration (mean 3 - 3.7 days) in SECURE trial subjects was generally consistent with approved low - dose CHCs ▪ Breakthrough bleeding also generally consistent with approved low - dose CHCs 2 and decreased during the 13 cycles of the study ▪ 2.2% of SECURE trial subjects discontinued due to a bleeding - related adverse event (Quartette: 5%; Natazia : 2.3%; Ortho Evra 1.1%) Bleeding Profile Similar to Oral Low - Dose CHCs 22 Analysis of subjects in the Safety Population of SECURE trial 1 - Moreau C, et al. Contraception 2007 ; 2 - publicly available product labels Mean Number of Unscheduled Bleeding/Spotting Days by Cycle

NASDAQ: AGRX ▪ 99.2 - 99.7% of patch adhesion scores ranged from no lifting of the patch (score = 0) to slight lifting at the edges (score = 1) — As rated by SECURE trial Investigators during routine subject visits, using a 5 - point scale provided by the FDA Adhesion Profile in the SECURE Trial 23 Investigator - Reported Patch Adhesion Scores Score 2 - 4 2: ≥ 50% adhered but < 75% (at least half of system lifts off) 3: < 50% (more than half of the patch lifts off, but the patch remains attached) 4: Patch completely detached Score 0 - 1 0: ≥ 90% adhered (none to minimal lift) 1: ≥ 75% adhered but < 90% (some edges showing lift) 88 90 92 94 96 98 100 1 2 3 4 5 6 7 Percentage of Scores Patch Day

NASDAQ: AGRX Observable Trend in Pearl Indices for Approved Combined Hormonal Contraceptives (CHCs) 24 Product (Year Approved) Pearl Index Ortho Evra (2001) 1.07 Nuvaring (2001) 2.02 Yasmin (2001) 0.4 Seasonale (2003) 1.98 Loestrin 24 FE (2006) 1.82 Seasonique (2006) 1.34 Yaz (2007) 1.41 Lybrel (2007) 2.38 LoSeasonique (2008) 2.74 Natazia (2010) 1.64 LoLoestrin (2010) 2.92 Generess (2010) 2.01 Quartette (2013) 3.19 Quartette ≥90kg 4.82 Quartette ≥70 to <90kg 3.38 Quartette <70kg 2.59 Sources: Trussell, et al., The Creeping Pearl (2013), currently marketed product labels, and publicly available information Quartette Overall Twirla Normal (BMI <25) Twirla Non - Obese (BMI <30) Twirla Overall (mean BMI 28.3) Twirla Overweight (BMI 25 to <30) Twirla Obese (BMI ≥30) Quartette ≥90kg Quartette ≥70kg to <90kg Quartette <70kg 0 1 2 3 4 5 6 7 2000 2005 2010 2015 2020 Pearl Index Historical Pearl Indices for CHCs Approved Since 2000 and the Pearl Indices Observed in the SECURE Trial Approved Combined Hormonal Contraceptives, CHCs Twirla Quartette Weight Groups Linear (Approved Combined Hormonal Contraceptives, CHCs)

NASDAQ: AGRX Twirla Demonstrates Non - Inferior Adhesion to Xulane in Comparative Wear Study 25 STUDY DESIGN ▪ Subjects randomized to wear either Twirla or Xulane for Week 1, then switched to the other patch for Week 2 ▪ Investigators assigned a daily patch adhesion score using a 5 - point scale from 0 (essentially no patch lift off skin) to 4 (complete patch detachment) ▪ Primary endpoint - mean difference in adhesion scores between Twirla and Xulane ▪ Non - Inferiority (NI) criterion – mean difference between upper 95% confidence limits must be less than +0.15 RESULTS: NON - INFERIORITY CRITERION MET . Patch Adhesion Score Range Note: Graph is for illustrative purposes only and is not to scale 0 (no lift) 4 (detachment) 0 Twirla 0.14 Xulane 0.39 Mean Adhesion Scores ;

NASDAQ: AGRX Comparative Wear Study 26 Study Design ▪ Study design is consistent with the 2018 ANDA Guidance document entitled Assessing Adhesion With Transdermal and Topical Delivery Systems for ANDAs ▪ Randomized, open - label, crossover adhesion study ▪ Healthy women aged 18 - 35 years with Body Mass Index (BMI) <35 kg/m 2 ▪ 77 subjects were included in the Per Protocol population used in the primary analysis ▪ Subjects were randomized to wear either Twirla or Xulane for the first week then switched to the patch not initially worn for the second week ▪ Investigators assessed patch adhesion for each day of wear and assigned the patch a daily score of 0 (essentially no patch lift off skin) to 4 (complete patch detachment) ▪ The mean score for Twirla (0.14) was numerically better than the mean score for Xulane (0.39), producing a negative mean difference ( - 0.25) ▪ No complete detachments of Twirla or Xulane occurred during the trial ▪ The upper bound of the 95% confidence limit for the mean difference is - 0.16, thus Twirla met the non - inferiority criterion of +0.15 Comparing the adhesion of Twirla to that of Xulane , the generic version of Ortho Evra POSITIVE TOPLINE RESULTS

NASDAQ: AGRX Contraceptive Use by U.S. Women 27 Any method 37.8M No method, at risk 4.4M No method, not at risk 19.3M Contraceptive Method Choice (Number of U.S. Women*) Number of U.S. women using a CHC method* (pill, ring, patch) 10,547,479 *In 2014 Source: Fact Sheet - Contraceptive Use In the United States, Guttmacher Institute, July 2018

NASDAQ: AGRX 28 Average Price Per Cycle for Branded CHCs ($WAC)
